 One of the big challenges in developmental neurobiology is to understand how the immense variety of neural types that constitute the nervous system is generated.
Multiple studies have shown the essential role of different signal transduction pathways in this process. And over the past few years it has also become apparent that
signal transduction pathways are not mere linear cascades. Conversely, they are organized into complex signaling networks. However,
the mechanisms by which signal transduction pathways within these networks are modulated and integrated are poorly understood.
One of the big challenges in developmental neurobiology is to understand how the immense variety of neural types that constitute the nervous system is generated.
Multiple studies have shown the essential role of different signal transduction pathways in this process. And over the past few years it has also become apparent that
signal transduction pathways are not mere linear cascades. Conversely, they are organized into complex signaling networks. However,
the mechanisms by which signal transduction pathways within these networks are modulated and integrated are poorly understood.
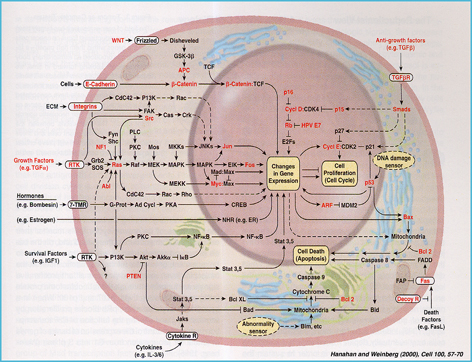
PDZ (PSD-95, Dlg, ZO-1) domain-containing proteins are excellent candidates as hubs of cross-talk between signaling cascades during Development. In addition, malfunction of many PDZ proteins has been linked to diverse neuropathologies and types of cancer. The aim of our research is to analyze the function of PDZ proteins, including the cytoplasmic protein Canoe/AF-6, as signal integrators within signaling networks during the acquisition of unique cell fates throughout nervous system development.
For this purpose, we are focused on the analysis of Asymmetric Cell Division, a fundamental and universal process to generate cellular diversity. Specifically we aim to answer the following questions:

1. - Which are the extrinsic and intrinsic mechanisms that regulate the asymmetry of the division to finally render two different daughter cells?
For answering this question we analyze the asymmetric division of embryonic and larval neuroblasts, the neural stem cells of the Drosophila central nervous system.
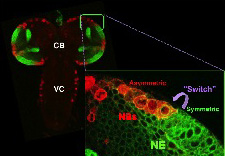
2. - Which are the extrinsic and intrinsic mechanisms that control the “switch” between a symmetric to an asymmetric mode of cell division?
Our model system for answering this question is the “Optic Lobe" of the Drosophila larval brain.
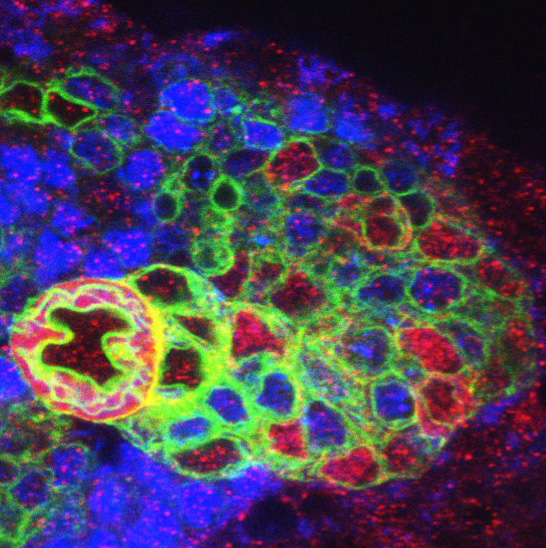
3. - Which are the connections between asymmetric cell division and cellular immortalization/tumorigenesis?
Our model system for studying these connections is the type II neuroblasts of the Drosophila larval brain.
We achieve our research integrating Genetic, Cell Biology, Biochemistry, Molecular Biology and Proteomic techniques.

